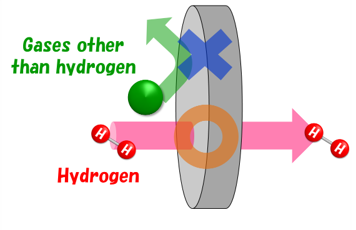
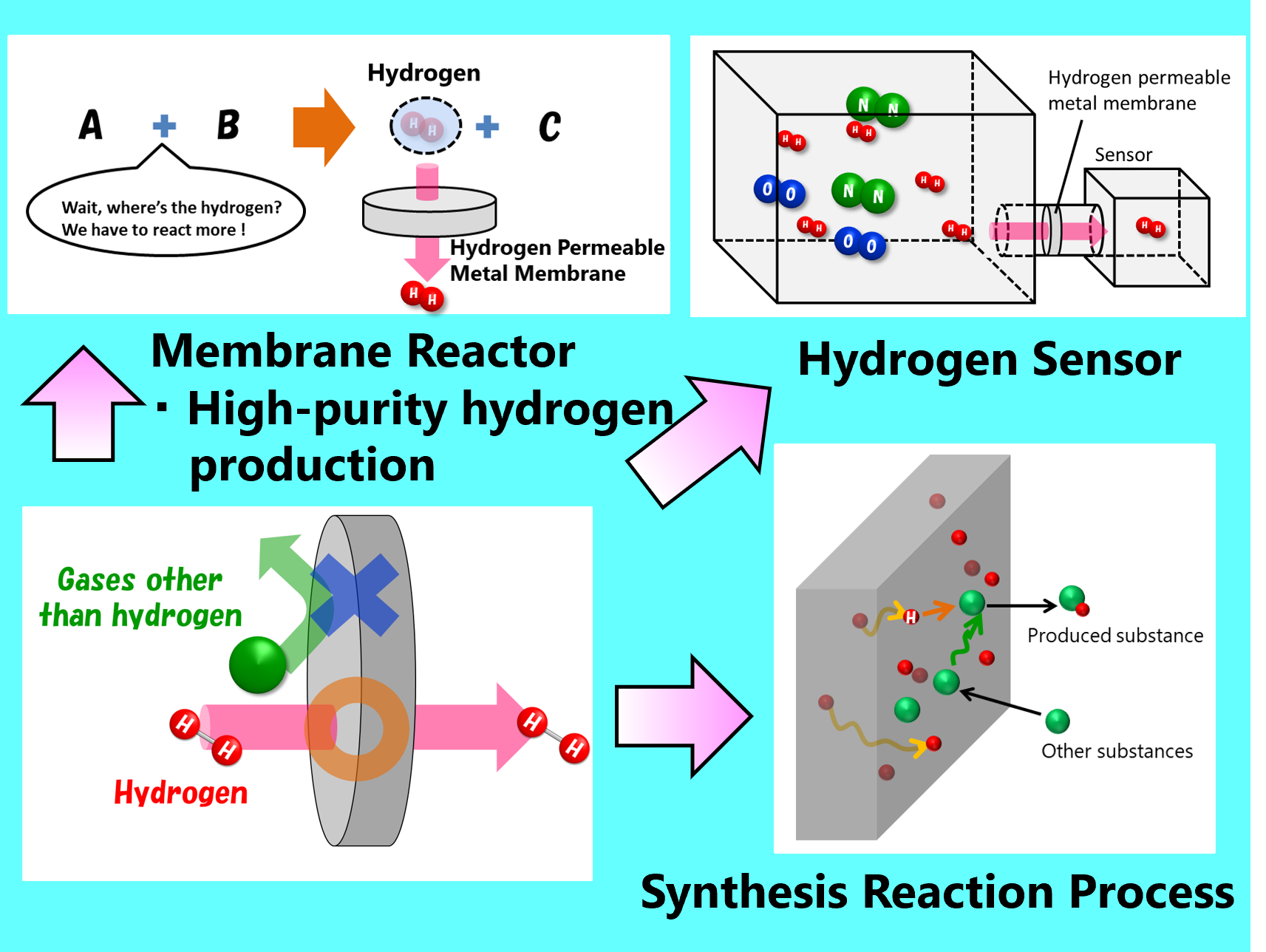
Hydrogen permeable metal membranes are mysterious membranes that allow only hydrogen to pass through, but not all metals have this property.
As of 2025, the most commonly used hydrogen permeable metal membrane is made of a metal called "palladium(Pd)".You may not have heard of this metal before, but it is a precious metal like gold and silver.
Since it was discovered about 150 years ago that palladium allows hydrogen to pass through, research has continued.
Here, I will explain a few terms.
First, a mixture of two or more metals is called an "alloy".
In addition, the ease with which hydrogen passes through a material is called "hydrogen permeability".
I will use these two terms in my explanation.
Alloys made by mixing palladium with silver (Ag) or copper (Cu) have even better properties than pure palladium.
For example, alloys of palladium and silver have higher hydrogen permeability than pure palladium.
Additionally, an alloy of palladium and copper, which contains approximately 50% copper, is cheaper than pure palladium and has the characteristic of being resistant to gases that palladium and palladium-silver alloys are not resistant to.

Palladium-based alloy membranes
The photos show alloys of palladium and silver, and palladium and copper, respectively. Although they look identical, their properties are completely different. This is one of the interesting aspects of metals and alloys.
However, there are several drawbacks to palladium and its alloy films.
1.Palladium is a very expensive metal, so it is not possible to produce a large number of hydrogen permeable membranes.
2.The hydrogen permeability of palladium is not sufficient to produce a large amount of clean hydrogen.
In order to overcome these shortcomings, there is a need to develop new hydrogen permeable metals that are inexpensive and have high hydrogen permeability.
Therefore, new hydrogen permeable metal membranes using niobium (Nb) and vanadium (V) have been developed.
Please refer to the table below.
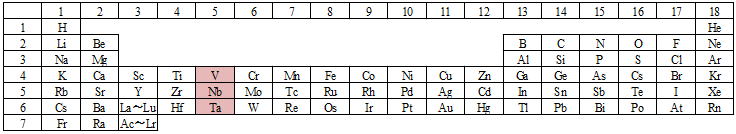
periodic table
This table is called the "periodic table" and summarizes the elements that exist in the world according to their similar properties.
Niobium and vanadium are in group 5 of the periodic table (the columns are called groups), so permeable membranes made from these metals are called group 5 metal hydrogen permeable membranes.
Group 5 metal hydrogen permeable membranes have high hydrogen permeability and are significantly cheaper than palladium. Based on this information alone, they appear to be the most effective option; however, unfortunately, they also have drawbacks.
Group 5 metals are highly compatible with hydrogen and absorb large amounts of it.
As a result, they undergo "hydrogen embrittlement", which causes them to deteriorate. Hydrogen embrittlement increases the risk of membrane failure.
Please refer to the figure below.

Hydrogen embrittled niobium
This image shows a niobium film that has cracked due to hydrogen embrittlement.
Although metals generally have good "ductility", the presence of hydrogen causes them to crack like this.
However, as it is, this material cannot be used as a hydrogen permeable membrane, so we are considering alloys to improve this shortcoming.
Various alloys have been developed, but our research group has developed alloys containing tungsten (W) and molybdenum (Mo).
These metals are in Group 6 of the periodic table mentioned earlier.
Alloying avoids hydrogen embrittlement, and as shown in the figure below,the material does not crack even after hydrogen has passed through.!

Niobium alloy
Furthermore, these alloys have hydrogen permeability approximately ten times higher than that of palladium, making them highly permeable to hydrogen. With further development, we may see the creation of even more remarkable alloys in the future.
 |
☆ Key points! Palladium is a typical material used in hydrogen permeable membranes, but recently, group 5 metals, which are less expensive and have superior hydrogen permeability, have been attracting attention. Group 5 metals are susceptible to hydrogen embrittlement, but alloying can prevent this. |