| ●Top | Hydrogen Permeable Metal Membrane | Category | Formula | Usage | Hydrogen Utilization |
 |
This page contains some difficult content, so feel free to skip it if you wish. A high school level understanding of mathematics is required to understand it. I have written it as simply as possible, so please take a look if you are interested. |
Now, in order to consider hydrogen permeability, which is the ease with
which hydrogen passes through, we need to consider why hydrogen moves (diffuses)
through the membrane.
To do so, please take a look at the figure below.
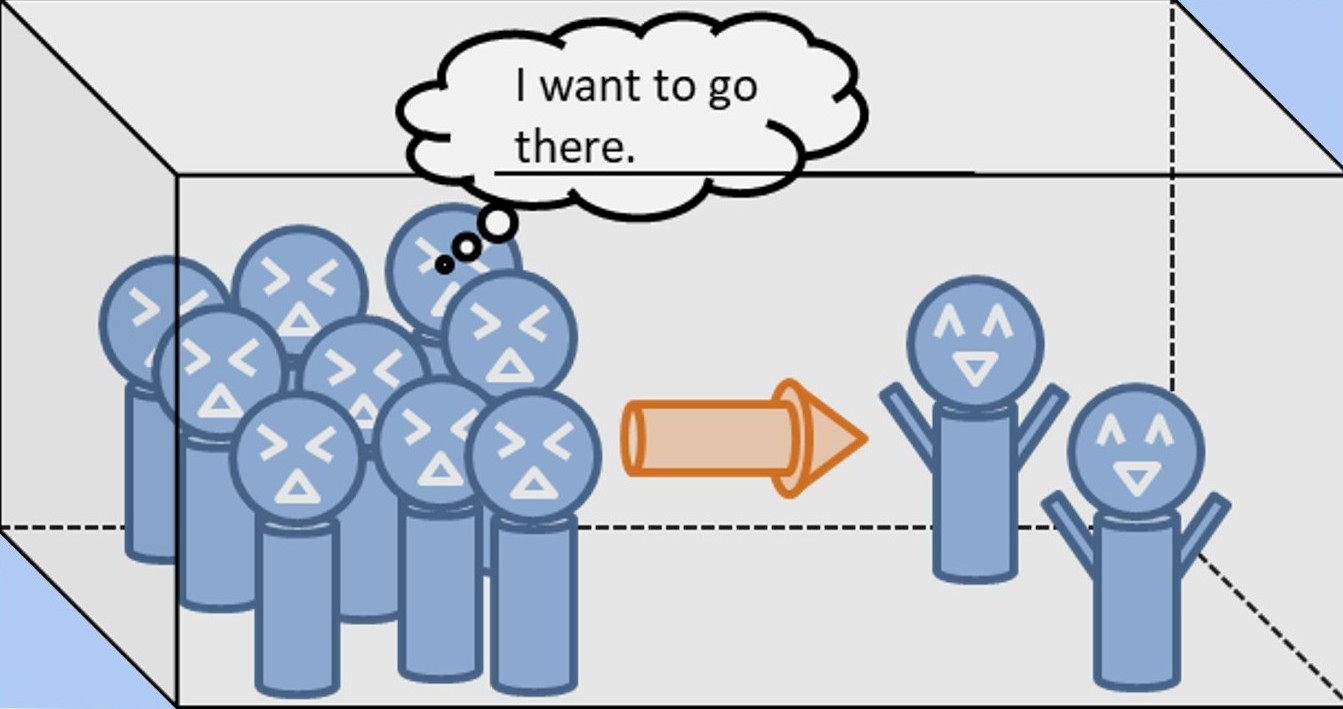
There are a lot of people crowded on the left side. This is very cramped and uncomfortable.
On the other hand, the right side is empty and very comfortable. In this situation, anyone would want to move to the right side.
Hydrogen also diffuses from the crowded side to the empty side in the same way.
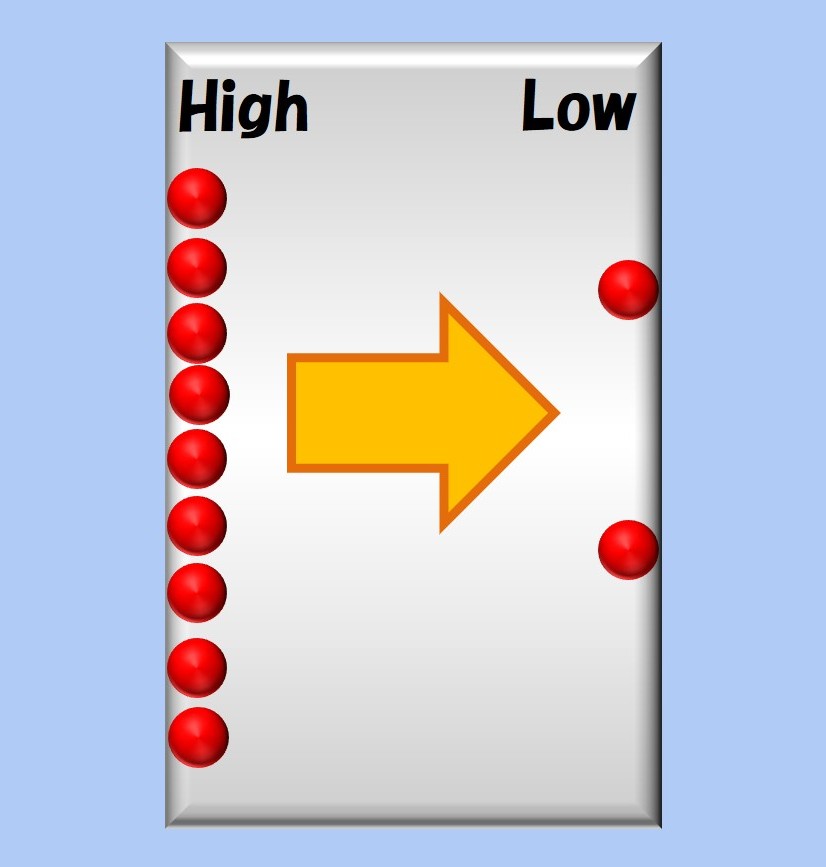
At this point, the more crowded the space on the left, or the more empty the space on the right, the more you will want to move to the right. This can be expressed as the following formula.
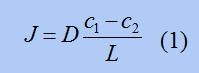
In this equation, J corresponds to the flow rate and is called "flux". c1is the concentration of the more concentrated side in the metal, and c2 is the concentration of the less concentrated side. This concentration is called "concentration". L is the thickness of the membrane. The thinner the membrane, the easier it is for the fluid to flow. This equation is derived from "Fick's law.
Now, this is the equation for hydrogen movement within the membrane. So why is the membrane filled with hydrogen atoms? It is because hydrogen gas dissociates into atoms and dissolves into the metal. ("What is a hydrogen permeable metal membrane?"). The following equation is said to apply to this dissociation and dissolution.
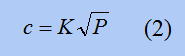
In this equation, c is the same hydrogen concentration as above, and P is the "pressure" of hydrogen, which can be thought of as the concentration of hydrogen in the gas. This equation is called “Geebels' law" and is an equation that links hydrogen in gas with hydrogen in metal. By inserting this equation into the above equation, the following equation is obtained.
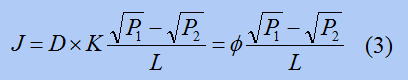
Here, φ is called the "hydrogen permeability coefficient" and is an indicator of how easily hydrogen passes through.
Looking at the equation, we see that φ=D×K. D is called the "diffusion coefficient" of hydrogen and represents how easily hydrogen moves within a metal. K is called the "hydrogen solubility coefficient" and represents how easily hydrogen enters a metal.
In other words, φis a combination of the ease of hydrogen movement and the ease of hydrogen penetration. Using φ we can compare the ease of hydrogen penetration through a metal film. People have investigated this φ for various metals.
This equation is certainly effective to a certain extent in expressing the permeability of hydrogen, but it is not perfect. In fact, there have been numerous reports of cases where this equation does not hold true. Please refer to the figure below.
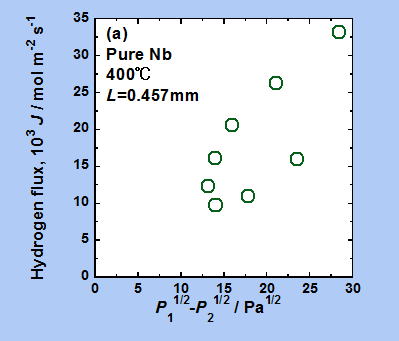
Analysis using conventional methods
This figure shows the experimental results when hydrogen was actually permeated through a metal film of Nb (niobium). If equation (3) holds true, J and √P1-√P2 should be proportional, and the graph should be linear. However, looking at this figure, it does not appear that the data points are arranged in a straight line. In other words, equation (3) does not hold true in this case.
Why does this not work in some cases? It is because Zibel's law mentioned earlier only applies when there is not much hydrogen present. When hydrogen actually passes through, there is a lot of hydrogen inside the metal, so it is impossible for the formula for when there is little hydrogen to work. Therefore, we propose the following formula.
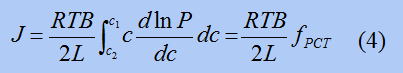
Well, I won't go into too much detail about this equation here, but at least it can express the phenomenon of hydrogen passing through better than the previous equation. First of all, of course, in this equation as well, the more crowded the hydrogen is, the more it will try to move. Furthermore, in this equation, in addition to the crowding of hydrogen, the discomfort of hydrogen is also taken into account. Please take a look at the figure below.
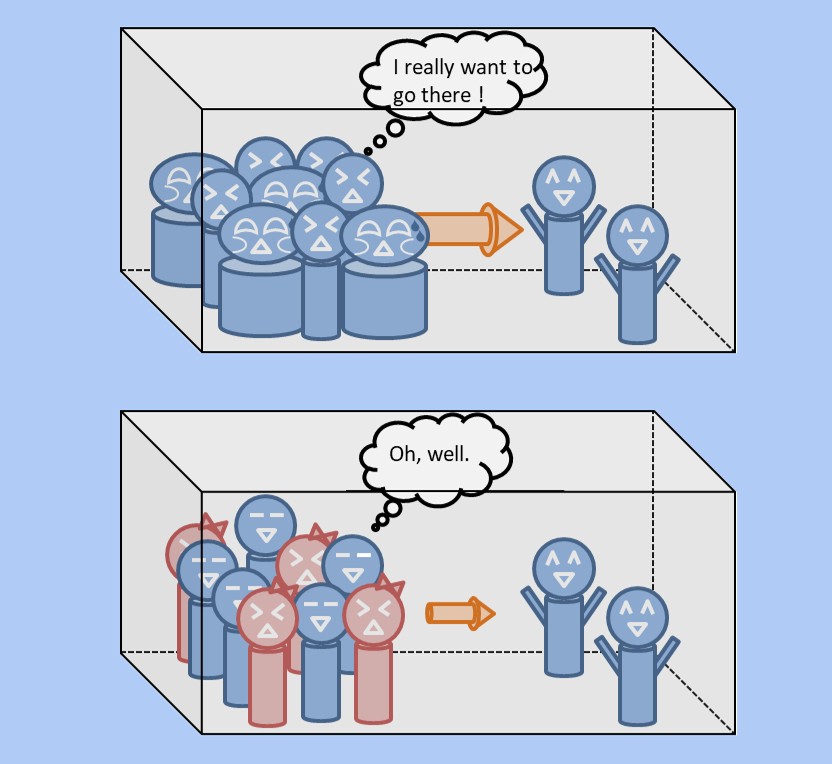
In this way, even if the crowd density is the same, the feeling of discomfort varies depending on the surrounding environment, right? If you are surrounded by strange old men, you want to move immediately, but if you are surrounded by beautiful women, the crowd density is not so uncomfortable. Hydrogen also has atoms that want to be around it and atoms that do not, and by incorporating such effects, the permeability of hydrogen can be better expressed. Please refer to the figure below.
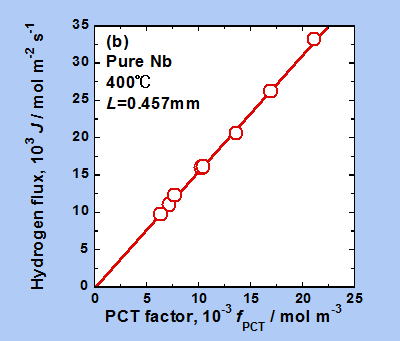
Analysis using a new analytical method
This graph shows the same results as the previous graph, which did not show a linear relationship, but by changing the horizontal axis, the points are now arranged in a very linear fashion. In this way, we can gain a deeper understanding of hydrogen permeability through the new equation.
 |
☆ key point! Hydrogen permeability is expressed by the hydrogen permeability coefficient φ, which is determined by the ease of hydrogen dissolution and diffusion. However, a new formula has recently been proposed that more accurately expresses hydrogen permeability. |